High Content Is Now High Throughput: End-to-End High Content Imaging and Subcellular Analysis of Autophagy in Minutes
Authors: Matthew Boisvert and Josh Kieler
Introduction: Measuring Autophagic Flux at High Throughput Speeds
- Cells eliminate intracellular waste and defective components through autophagy (“self-eating”)
- A key therapeutic target for aging-related dysfunction, autophagy is implicated in neurodegeneration and cancer
- Autophagic flux can be measured by quantifying autophagic vesicles, visualized through examining marker protein LC3b or dyes activated by vesicle-specific conditions
- High content imaging (HCI) allows for cell-level resolution, visualizing aggregates and defining cell borders, quantifying spots per cell
- Resolving aggregated autophagosomes is essential, cell lines often have baseline levels of autophagy, generally non-aggregated
- Screening large compound libraries for autophagic flux requires speed, low variability and accurate detection
- Using the Araceli Endeavor® HCI Platform this assay demonstrates:
- Assay validation with four mechanistically different compounds
- Submicron resolution to accurately visualize aggregates
- Elimination of variability with whole well imaging
- <10-minute scan times for 96, 384 and 1536-well high content plates
- Flexibility: equivalent results with immunohistochemistry (IHC) and cell dye-based workflows
Materials and Methods

Figure 1: high content assay, imaging, and analysis workflow for a single plate
Assay: Human bone carcinoma (U2OS) cells were plated at 12500 cells/well in 96 well Grenier CellView plates and cultured for 48 hours in standard conditions (complete media, 5% CO, 37°C). Cells were treated with compound or DMSO in a 1:2 dilution series, 3 replicates/plate, for 12-16 hours.
Treatments: DMSO: vehicle control; [hydroxy]chloroquine: autophagosome degradation inhibitor; verapamil: autophagy inducer via Ca2+ inhibition; MG-132: autophagosome arrest via proteasome inhibitor; torin-1: autophagy inducer via mTOR inhibition
lmmunohistochemistry-based detection and analysis: Cells were fixed in 10% formalin for 15 minutes, permeabilized with 0.1% Triton x-100, then stained and counterstained for 60min/step in 1% BSA, with 1:1000 Rb anti-LC3b (L10382) and 1:1000 Gt anti-Rb Alexa 488 with 1:750 Hoechst 33342 (nuclei), and 1:1000 iFluor Phallodin-647 (actin). Cells were washed 4x between steps and all solutions are in PBS pH 7.4.
Dye-based detection and analysis: Cells incubated at 37°C with 1:500 dilution of cationic amphiphilic tracer (CAT) CYTO-1D®Autophagy detection kit 2.0 in complete media, then fixed (10% formalin for 15 minutes) before staining with 1:750 Hoechst 33342.
Imaging: The 96-well plate was imaged on Araceli Endeavor®, in 2 channels (live cell) or 3 channels (IHC), with full well (4.5mm x 4.5mm/well), submicron resolution (.27µm pixel size). Data (3072-7200 images) collected for the whole 96-well plate in 4-10 minutes.
Analysis: Araceli internal analysis tool used for all analysis. For both assays, machine vision template match used to quantify nuclei and autophagic vesicles, with a larger template used for CytolD, as staining appeared more aggregate-like. For IHC data, nuclei used to seed cell, thresholded actin staining used to define cell, and cytoplasmic vesicles counted per cell (10-20 minutes). Live stain data analyzed segmentation-free, normalized to nuclear count (5-10 min).
Full Well, Full Plate High Resolution Scans in under 10 minutes
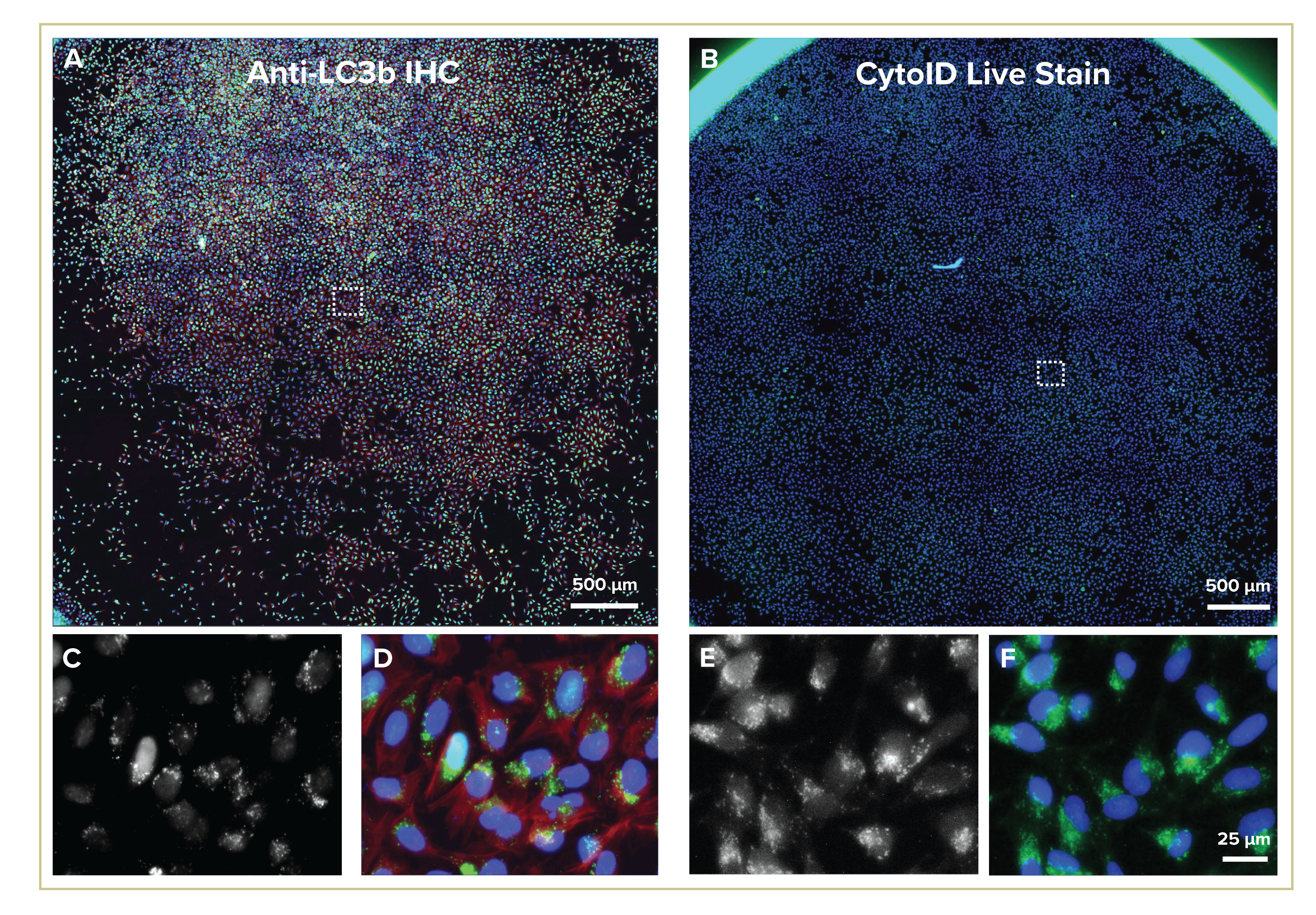
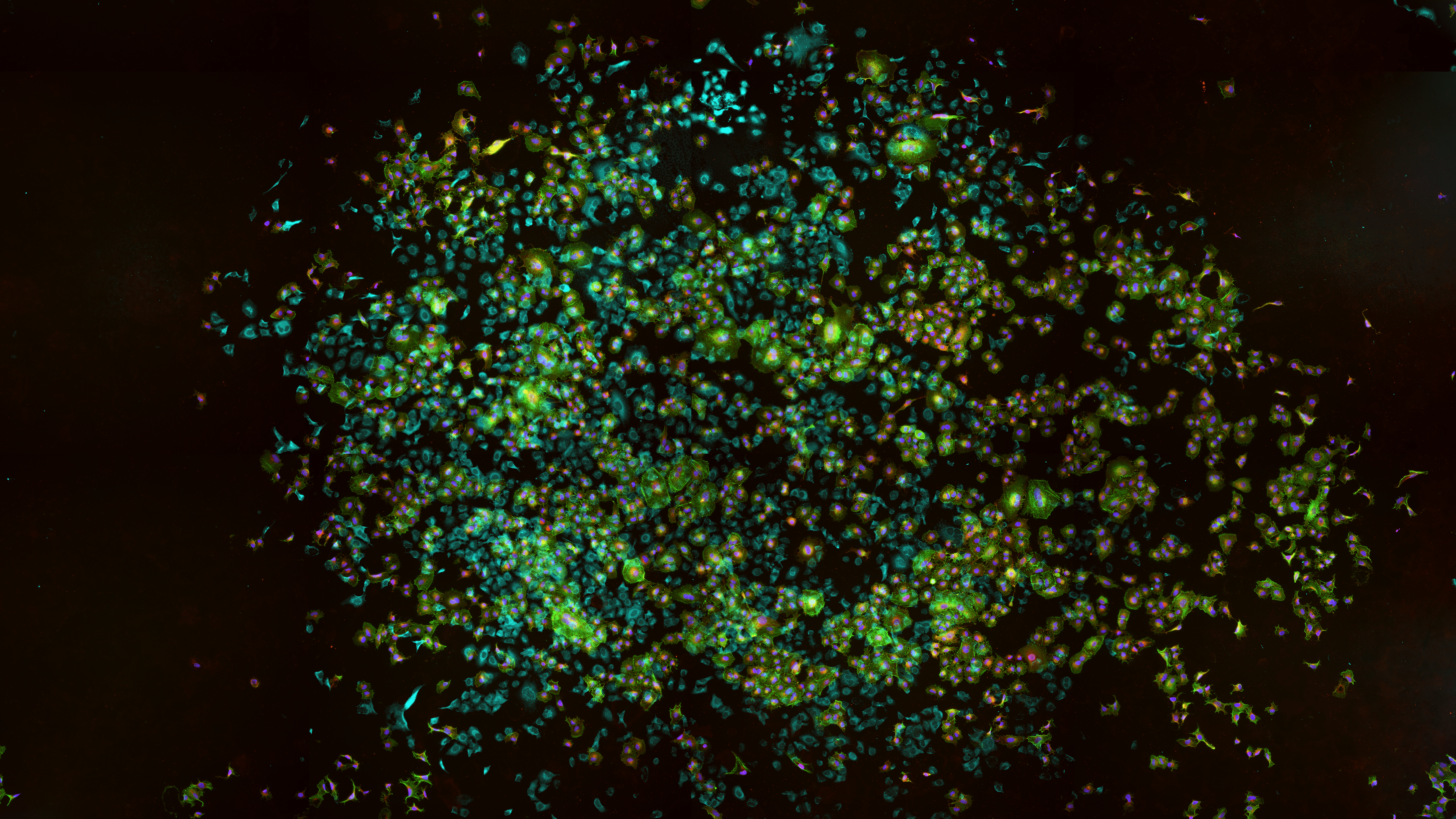
Figure 2: 96 well plates imaged at 0.27µm/pixel with full wells (>85% of well area) collected. 10µM chloroquine treatment shown. A) 3000×3000 pixel images with 5×5 fields of view/well for anti-LC3b antibody staining (green, actin in red and nuclei blue); B) 4400 x 4400 pixel images with 4×4 FOV/well taken for CytoID live stain (green, nuclei in blue). Zoom in as indicated by box with (C, E) autophagy stain and (D, F) merged image with counterstain.
Autophagic Flux Consistently Detected with Both Immunohistochemical and Live Cell Dye Assays
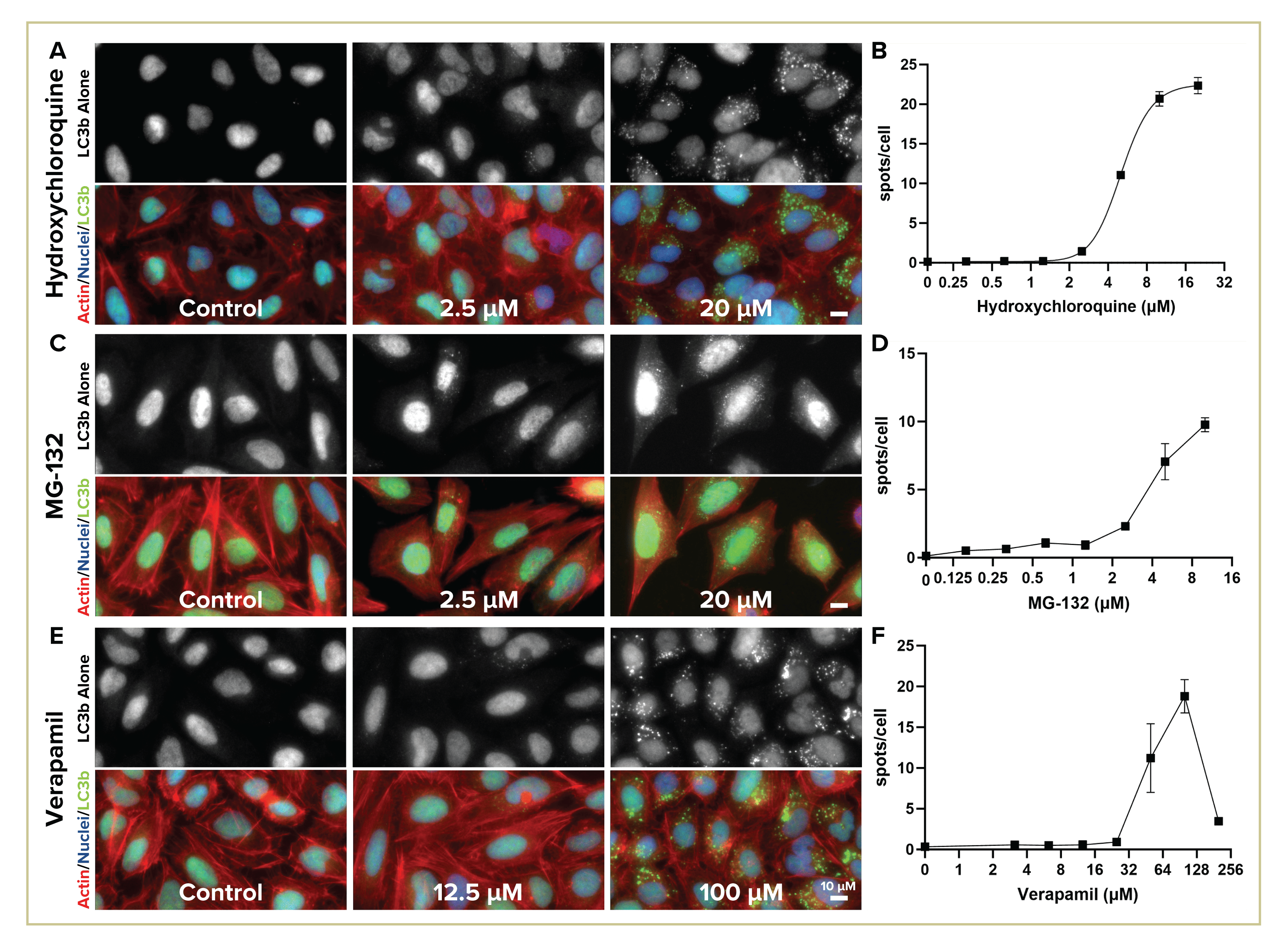
Figure 3: Immunohistological detection of Autophagic flux with LC3b antibody.
Autophagic flux consistently detected with both immunohistochemical (Figure 3) and live cell dye (Figure 4) assays. Representative images (A, C, E) of high, medium and control (no) dosages shown with autophagic vesicle staining (gray, upper panel) and merged with nuclear (both stainings) and actin counterstains (IHC only). Graphs of quantified staining (B, D, F) with nonlinear fit calculated for [hydroxy]chloroquine (B) and torin 1 (4D) with R2>0.98; EC50 for chloroquine is 5 µM in IHC and 3.5 µM for live dye experiment, Torin 1 EC50=0.1 µM (GraphPad Prism). Error bars +/- standard deviation. Verapamil (3E, F) is cytotoxic at high dosage.
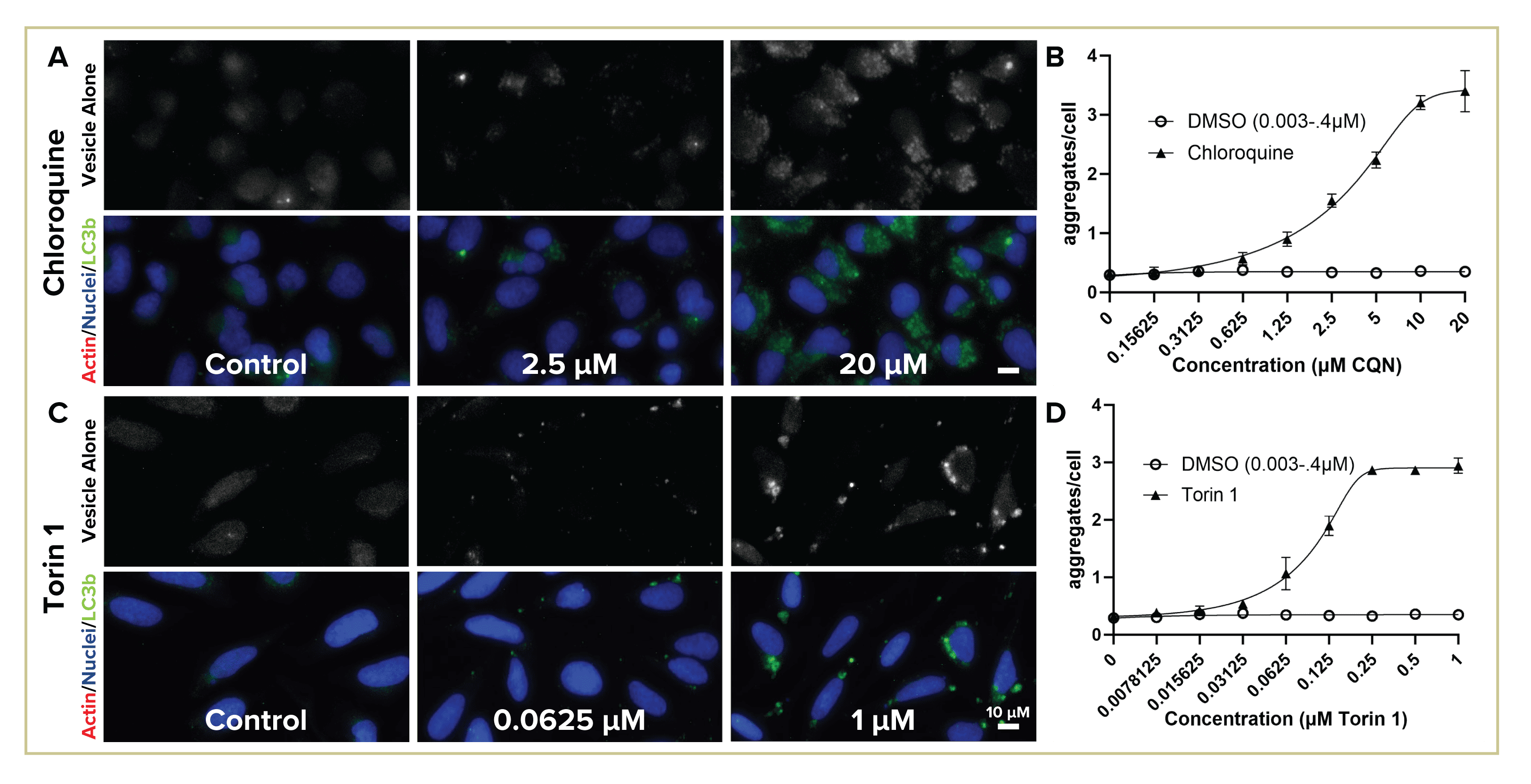
Figure 4: Detecting autophagic flux with live cell CAT dye.

Table 1: Z’ from screen data (above) using maximal response; IHC uses hydroxychloroquine.
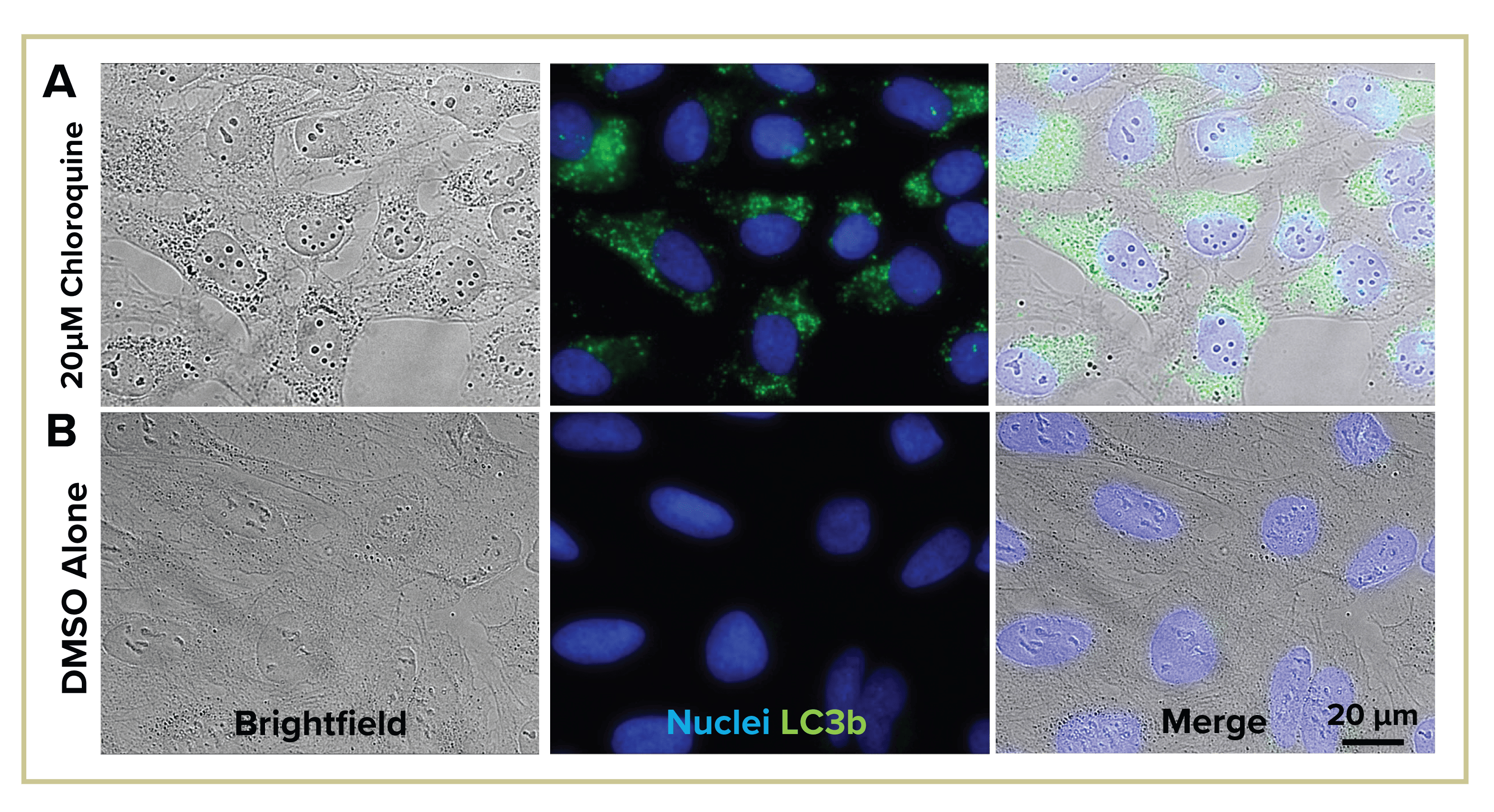
Figure 5: Morphological changes after autophagic flux visualized in brightfield. Adding only 2-3 minutes to a full well scan, changes in vesicle morphology and nucleoli visible in brightfield (i) after chloroquine treatment (A) compared to control (B). Darker, larger vesicles seen in treatment group, corresponding to LC3b stain (ii and iii).
Conclusion: High throughput high content imaging at submicron resolution with subcellular analysis
- Autophagic flux reliably detected using distinct modulators in dilution
- Consistent Z’>0.5 indicates robust effect size, minimal variability
- Plate to results delivered in under 30 minutes
- Assay reliability and flexibility at speed: both immunohistochemistry and live cell dyes yield consistent results with similar EC50 values
- 0.27µm pixel size allows for spot detection at submicron scale
- Autophagic vesicles visible in brightfield, allowing label-free detection
- Assay broadly generalizable to other quantitative spot-based HCI assays such as: subcellular protein localization, plaque formation, aggregation, FISH, phagocytosis
References
Djajadikerta A, Keshri S, Pavel M, Prestil R, Ryan L, Rubinsztein DC. Autophagy Induction as a Therapeutic Strategy for Neurodegenerative Diseases. J Mol Biol. 2020 Apr 3;432(8):2799-2821. doi: 10.1016/j.jmb.2019.12.035.
Joachim J, Jiang M, McKnight NC, Howell M, Tooze SA. High-throughput screening approaches to identify regulators of mammalian autophagy. Methods. 2015 Mar;75:96-104. doi: 10.1016/j.ymeth.2015.02.002
Sharifi MN, Mowers EE, Drake LE, Macleod KF. Measuring autophagy in stressed cells. Methods Mol Biol. 2015;1292:129-50. doi: 10.1007/978-1-4939-2522-3_10.
Marinković M, Šprung M, Buljubašić M, Novak I. Autophagy Modulation in Cancer: Current Knowledge on Action and Therapy. Oxid Med Cell Longev. 2018 Jan 31;2018:8023821. doi: 10.1155/2018/8023821